Drug Loading Impact on Release Kinetics
Microstructure Analysis of a Polymer Implant for Sustained Delivery of Meloxicam

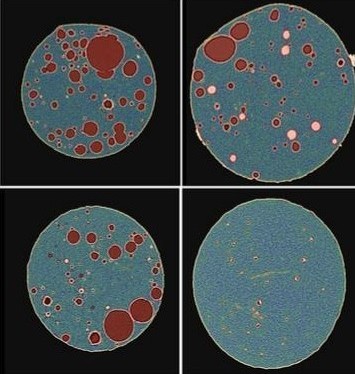
In order to effectively prevent subacute inflammation and prevention of biofouling formation, the Yeo Lab at the Purdue University College of Pharmacy developed a meloxicam-releasing polymeric implant. Three types of polymer matrices were loaded with meloxicam and drug release was investigated using solid-state characterization and image analysis. DigiM’s patented I2S software was used to reconstruct X-ray MicroCT images of the PCL matrices loaded with varying drug loads. Artificial intelligence segmentation classified the different phases to quantify their size and distribution including the PCL polymer, pores, and meloxicam clusters; additionally, a 3D reconstruction of the dataset was created to further visualize these segments. The ability to visualize the internal structure of the PCL matrix was important in understanding how changing the drug loading impacts the release kinetics and performance.

The Yeo Lab’s findings comparing all matrices showed that PLGA and EVA had limited control over drug release rate, respectively due to the acidic microenvironment and hydrophobicity of these polymers. It was seen that the PCL matrices showed more ideal meloxicam release, as demonstrated by DigiM’s image analysis of pore network development. Through imaging of pre and post release samples, the evolution of the microstructure features was correlated to the increased rate of drug release observed in vitro. This study suggests that the type of polymer matrix combined with the amount of drug loading is essential in controlling the drug release rate.
Additional Case Studies
Our Expertise In Numbers
Programs Supported
Formulations Digitized
Pharmaceutical Partners

Transform Your Program with Microstructure Science
Get started with a drug product digital twin.