Long-acting Injectables
Optimize long-acting injectable development with microstructure insights. Leverage in silico prediction for streamlined product optimization.

Issue
The performance of long-acting injectable systems designed for controlled release are dictated by the arrangement of polymer, porosity, and active ingredient. When these factors are ignored and treated as a black box, it extends the costs needed for development. This is tied to the lengthy formulation-release testing cycle, where the feedback loop for formulation design can rely on weeks to months of in vitro testing.
Common Challenges
- No method to quantify porosity and API properties within the microsphere
- Limited ways to elucidate the release mechanism and polymer behavior
- Lengthy development cycles due to long in vitro testing
- Lacking methods to demonstrate Q3 microstructural equivalence
Solution
We encourage our partners to formulate and manufacture through a quality by design approach, using the internal microstructure arrangement for faster decision making. This includes in silico release prediction based on characterized structures, enabling a rapid exploration of the formulation and manufacturing space.
- Internal 3D quantification of porosity and API quality attributes
- In silico release prediction to shorten formulation testing cycles
- Quantitative batch uniformity analysis for scale up and quality assessment
- NDA and ANDA data package support

Our Workflow
Our microsphere solution applies correlative microscopic analysis with FIB-SEM and X-ray Microscopy, ensuring detailed microstructure assessment and quantification of batch representativeness. Through image-based in vitro release simulation, formulation performance can be rapidly assessed, replacing and reducing the need for lengthy in vitro tests. Quantified microstructure data and release profiles can be used in NDA and ANDA filings to demonstrate understanding of drug product quality attributes and Q3 bioequivalence and quality. We support all microencapsulation systems, including beads, microparticles, and microcapsules.
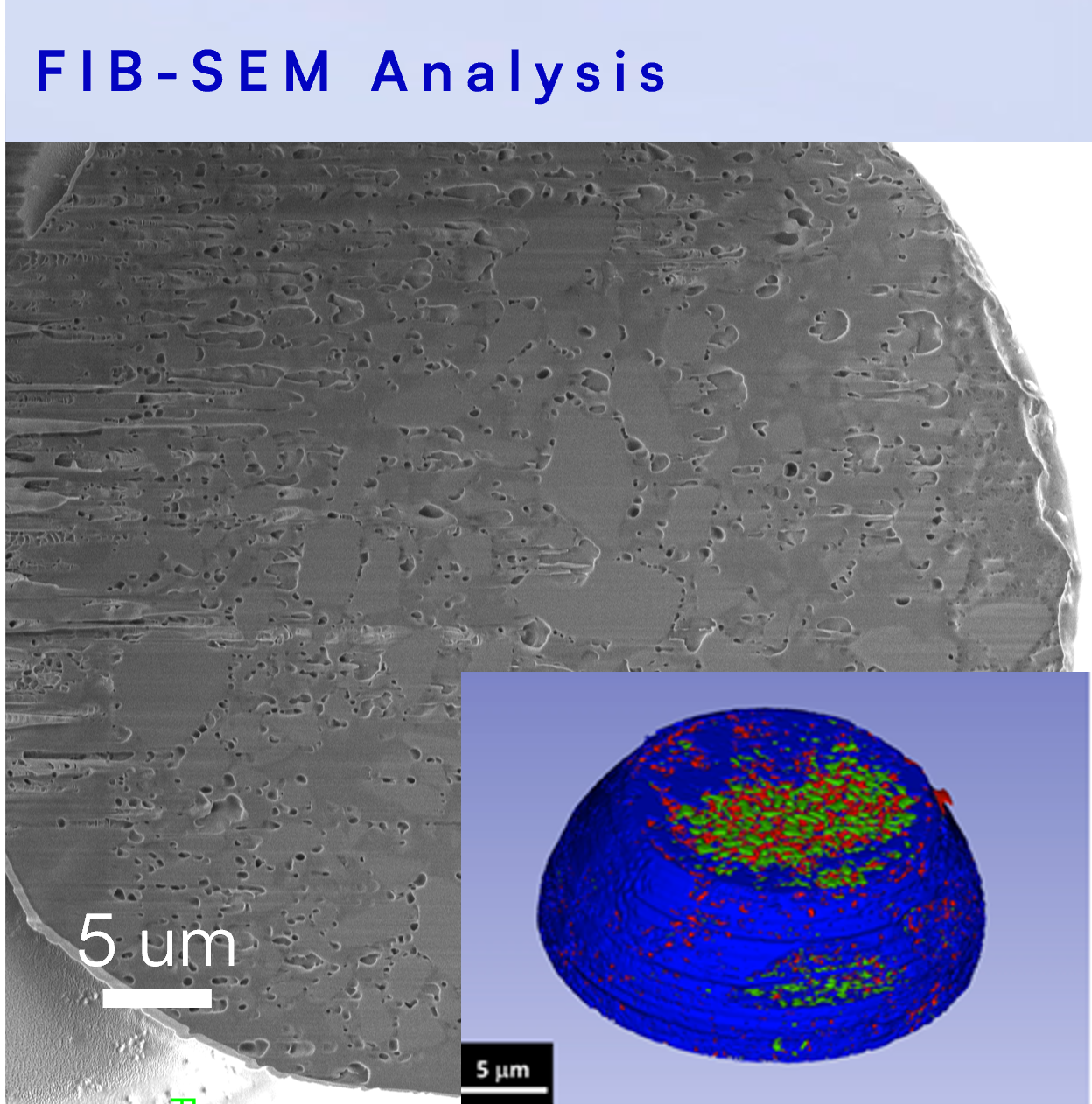
FIB-SEM analysis enables high-resolution measurement of porosity and drug domain attributes within the final product. Combining precise cross-sections with nanometer SEM resolution, FIB-SEM is one of the most robust approaches for true internal understanding of polymeric drug products and intermediates.
Other FIB-SEM applications include implants, IUDs, controlled release coatings, beads, and in situ formed implants.

Our true density measurement is a patent-pending method for quantification of material true densities and quantitative comparisons of drug product microstructure arrangement (US20240003797A1). Built for X-ray micro-CT and X-ray microscopy scans using specialized algorithms and a custom calibration system, our approach provides quantiative assessment for microstructure uniformity within and across batches.
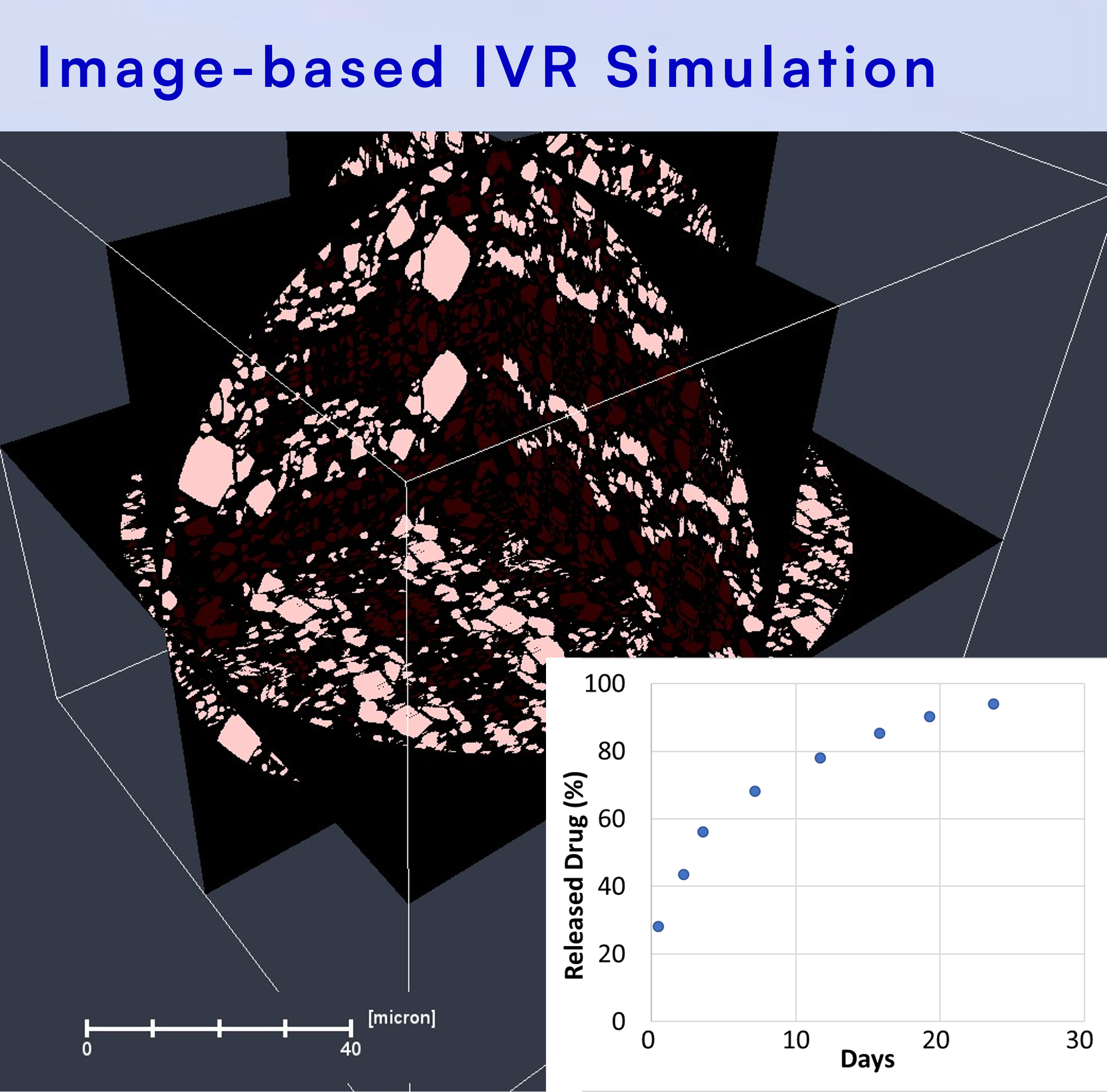
Our patented image-based simulation integrates the drug product microstructures for accurate predictions of in vitro drug release. Our simulation technology supports a variety of polymer systems and release modes, including diffusion, bulk and surface polymer erosion, and core-sheath.
Areas of Support
Process & Formulation Impact
Understand how manufacturing approaches, drug load, and polymer selection impact the internal microstructural arrangments that dictate release.
ICH Stability and Aging Tests
Study ICH stability exposed samples to evaluate product quality risk, including porosity and polymer evolution upon humidity and thermal exposure.
Optimize Particle Engineering
Perform rapid parameter navigation with sGAN structure synthesis, where the impact of particle size and drug load can be explored with digital microstructure engineering.
Reduce & Replace In Vitro Testing
Comparative microstructure studies of reference products, in house formulations, and SUPAC modified products.
Demonstrate Q3 Bioequivalence
Comparative microstructure studies of reference products, in house formulations, and SUPAC modified products.
Release Mechanism and Polymer Behavior
Examine post-partial release samples from in vitro or ex vivio tests to understand polymer behavior, degradation, and drug release.
Our Expertise In Numbers

Transform Your Program with Microstructure Science
Get started with a drug product digital twin.


