Capsule Batch Performance Differences
Assessment of In Vitro Release Testing Performance Differences

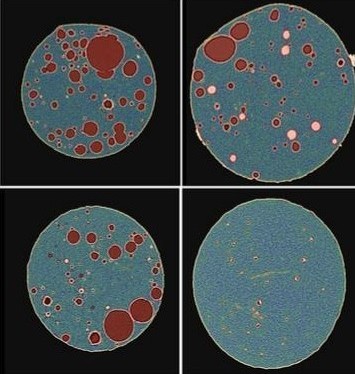
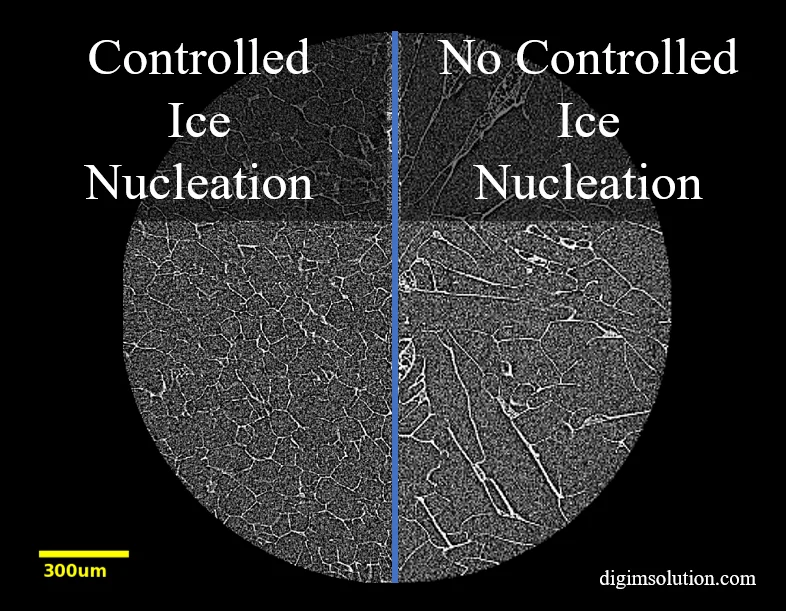
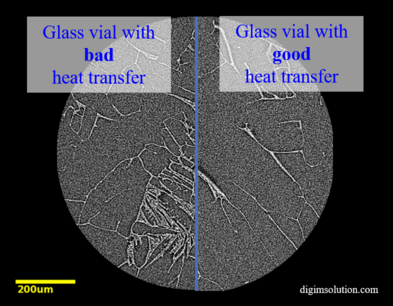
Particle morphology and particle size distribution are critical quality attributes (CQAs) important to characterize during the development of pharmaceutical products. In particular, the control of active pharmaceutical ingredient (API) particle characteristics is essential for optimizing dissolution and release performance. An understanding of the particle size distribution, shape, and spatial relationship is important for interpreting in vitro release testing differences and root causes of failure.

However, there is a striking lack of suitable approaches to investigate the size and distribution of API particles within the final product. In this project, we applied X-ray microscopy and artificial intelligence (AI) image segmentation to quantify the critical quality attributes of API particles. Through this quantitative analysis, new mechanistic understandings of drug release mechanisms were uncovered, and the root cause of in vitro release testing differences was discovered.
Additional Case Studies
Our Expertise In Numbers
Programs Supported
Formulations Digitized
Pharmaceutical Partners

Transform Your Program with Microstructure Science
Get started with a drug product digital twin.