digiM Patents Density Measurement Technology for micro-CT

digiM Awarded Patent for True Density Measurement from CT Images
Patented under US20240003797A1 "Method For Density Measurement Of Materials Using Computed Tomography With Diffraction Artifact Correction", the technique brings forth a significant asset to quantitatively measure material and sample true densities, directly from micro-CT data. By integrating density analysis with microscopic imaging data, the method provides a connection between density and microstructure attributes which cannot be discerned with traditional approaches like gas pycnometery or gradient density columns.
True Density Analysis for Microstructure Quality Comparison Across Batches
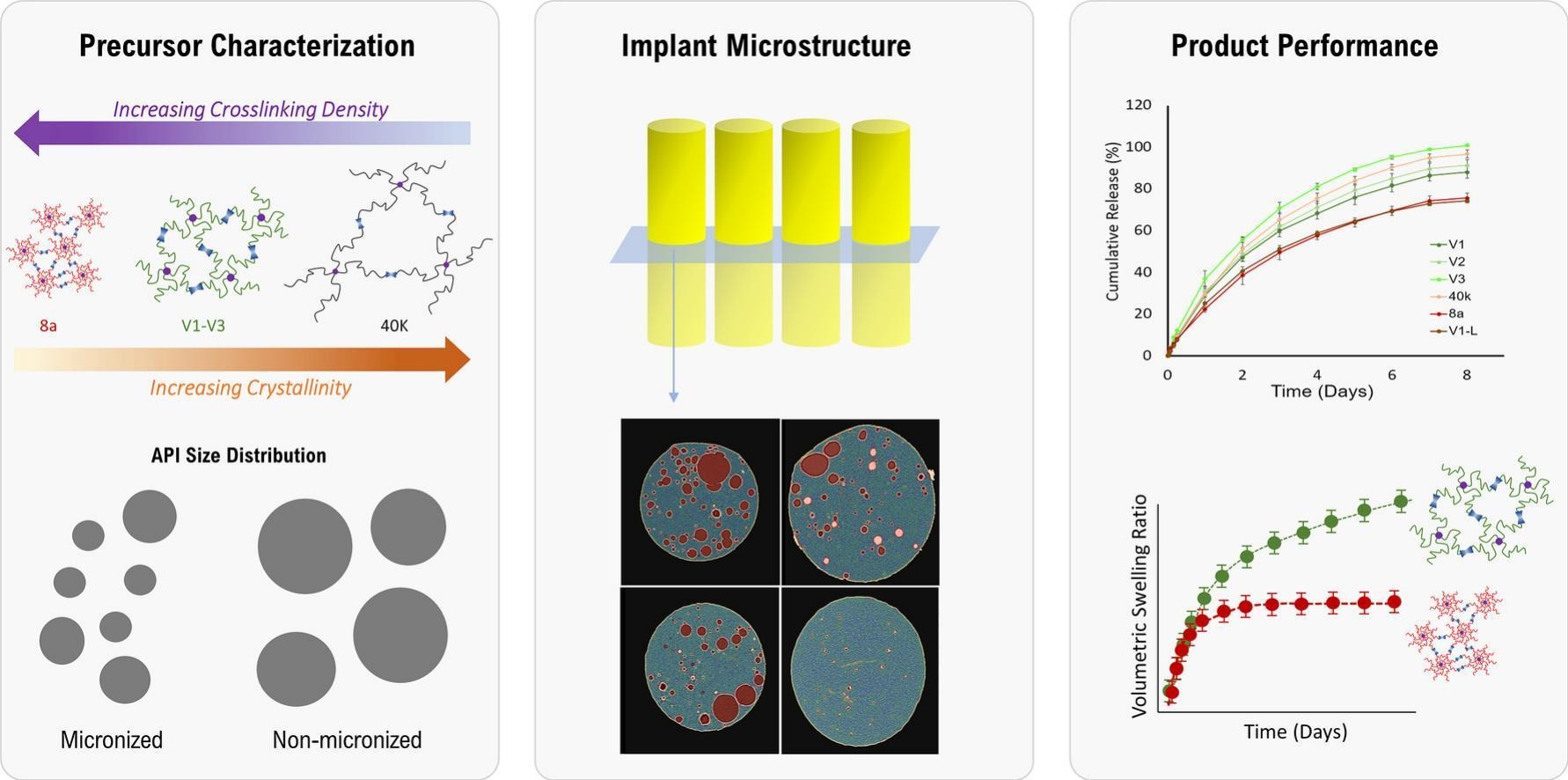
The patented method enables direct measurement of material true densities to compare individual samples (eg tablets), and uniformity within and across batches (eg powder lots). Applied to pharmaceutical and material science development, the technique enables quantitative analysis to evaluate:
- Batch Equivalence and Uniformity
- Aging and Stability Impact on Quality
- Product Quality
- Sub-Resolution Features
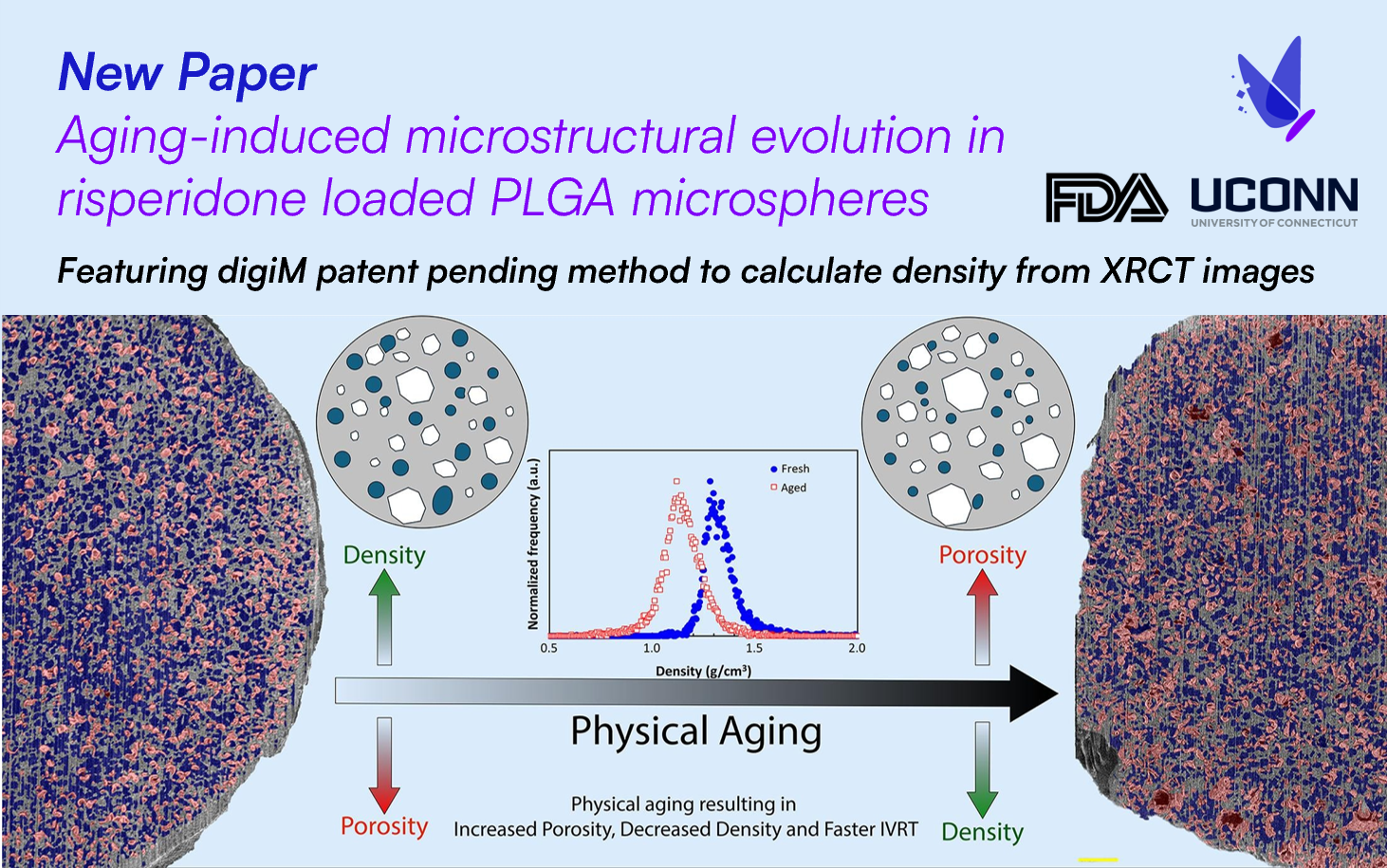
The innovation was first demonstrated in the paper "Aging-induced microstructural evolution in risperidone loaded PLGA microspheres." In this work, FDA researchers evaluated the usage of the technique to compare microsphere densities for fresh and aged batches. The results demonstrated how density analysis discerned a decrease in density of microspheres upon aging, altering the corresponding drug release rates. Since this publication, the density technique has been applied to dozens of pharmaceutical programs, including a quality by design approach for the design space for granule processing.
Platform Available for Projects and Licensing
digiM’s density measurement technique is available through service projects and in licensing by analytical teams running CT scans. A module within the digiM I2S platform, the technology is naturally integrated into the lifecycle of microscopic data management and analysis. The technology is available for researchers to implement within their internal quality checks, scale up assurance, ICH stability testing, and dissolution performance troubleshooting. Combined with CT data collection, density analysis provides a more robust pathway for measurable comparison and understanding on product quality and performance attributes.
Transform Your Program with Microstructure Science
Get started with a drug product digital twin.