digiM Co-Authors FDA Study on Dissolving Microneedle Quality

digiM Solution is proud to announce our co-authorship on a newly published paper in the International Journal of Pharmaceutics, alongside researchers at the FDA's Office of Pharmaceutical Quality (OPQ).
Microneedles are patch-based delivery devices with a needled surface where each needle is just microns in size. The technology has existed for years, primarily in cosmetics, but the pharmaceutical world has long seen its potential for something far more significant: bypassing the stratum corneum, the skin's outermost defensive layer, to deliver drugs directly into the subcutaneous space in a controlled, painless way. For drugs that degrade in the GI tract, for vaccines, and for women's health applications in low and middle income nations, microneedles represent a genuinely exciting delivery platform.
The challenge is that not a single microneedle drug product has received FDA approval. Manufacturing at the micron scale rules out traditional metal needles, and while polymers have emerged as the leading material candidate, critical questions remain unanswered: which polymer, what concentration, and how should the drug actually be released? Those open questions are exactly what this research set out to address.
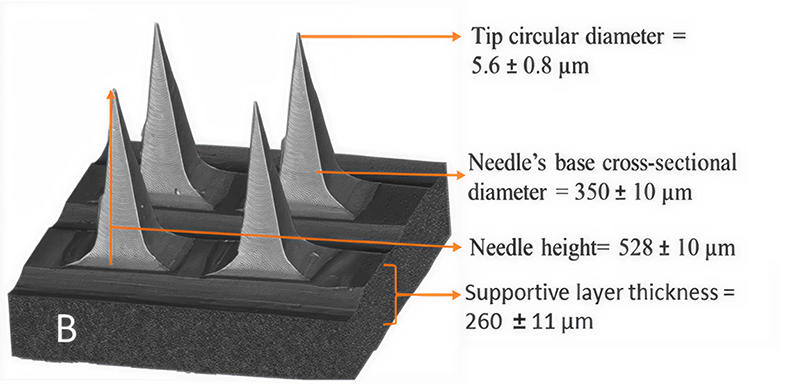
The study focuses on dissolving microneedles made from polyvinyl alcohol (PVA) and examines how the ratio of polymer to drug affects critical quality attributes including mechanical strength, drug distribution, and release behavior. digiM's contribution centered on AI-assisted image analysis using digiM I2S software, applying supervised machine learning to high-resolution X-ray microscopy and synchrotron nano-CT images to map drug distribution within the microneedle structures. It was a focused contribution, but one that surfaced meaningful insights into how drug localization within a microneedle influences how that drug ultimately permeates through skin.
Key findings from the study include:
- Higher PVA content increases structural integrity but slows drug release, presenting a direct formulation tradeoff
- Drug distribution within the microneedle structure correlates with permeation behavior across skin tissue
- Polymer content does not affect skin insertion capability or needle disintegration, pointing to selective impacts on specific quality attributes
- Membrane-free dissolution testing effectively differentiated between formulations, offering a practical approach to standardized quality control
This work is a step toward closing the quality and regulatory gaps that have kept microneedle drug delivery from reaching patients. We are proud to have contributed to that effort alongside the FDA.
Read the full paper: https://doi.org/10.1016/j.ijpharm.2026.126830
Transform Your Program with Microstructure Science
Get started with a drug product digital twin.