Synchrotron microCT Analysis for Spray Dried Particles

Synchrotron computed tomography combined with AI-based image analysis for the advanced characterization of spray dried amorphous solid dispersion particles
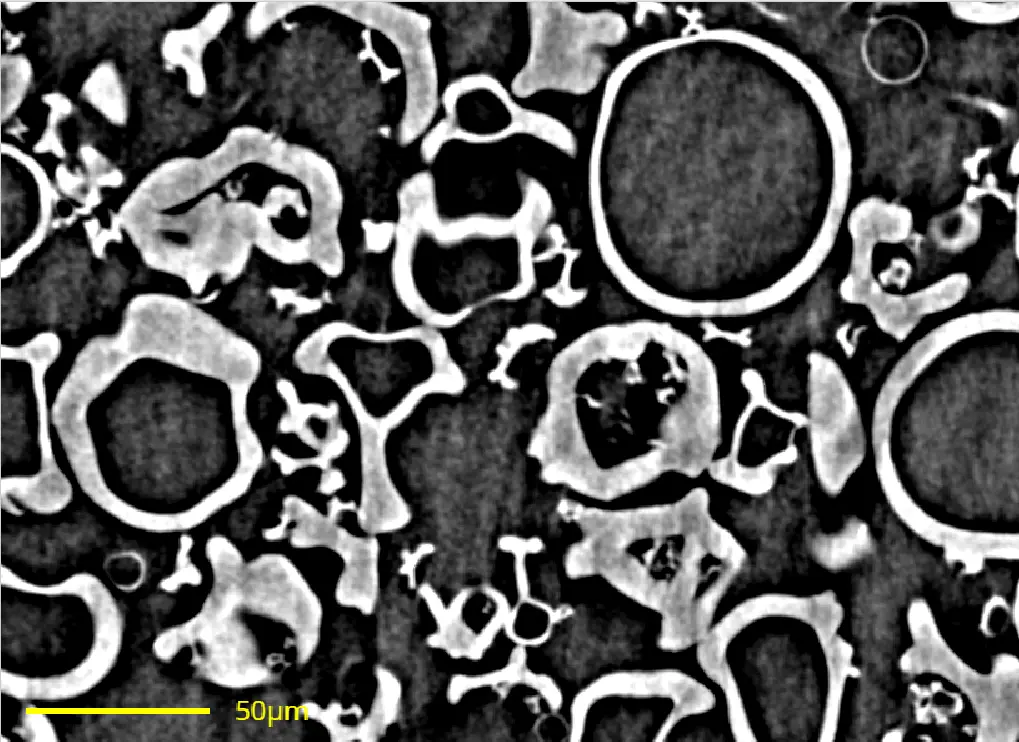
Particle engineering aims to design particles with specific properties. A deeper understanding of how particle formation relates to material attributes and process conditions are critical to strengthen knowledge on powder properties and enhance modeling capabilities. New, alternative powder characterization techniques can offer novel and more accurate measures for particle properties, giving more advanced characterization information. In this context, a case study is presented in which spray dried amorphous solid dispersion powders produced by modifying process conditions were characterized by both well-established compendial methods (i.e., laser light diffraction, SEM image analysis, bulk and tapped density, and gas adsorption), as well as a new method combining synchrotron computed tomography (SyncCT) with AI-based image analysis. SyncCT was used to classify and quantify the spray dried particles as hollow spheres and solid particles, giving a more detailed quality measure of the particle shape, as they impact downstream processing differently. Moreover, hollow particle wall thicknesses, as well as internal and external particle surface areas were measured by SyncCT. Altogether, powder characterization data from SyncCT show similar trends to that obtained from compendial techniques and giving additional quality measure regarding particle shape, showing promise of this new and advanced characterization method.
Tatiana Marcozzi, Sruthika Baviriseaty, Phillip Yawman, Shawn Zhang, Chris Vervaet, Valérie Vanhoorne, Sune Andersen
with Janssen and Ghent University
https://doi.org/10.1016/j.xphs.2024.10.033
Additional Publications
Transform Your Program with Microstructure Science
Get started with a drug product digital twin.














