Revealing Impact of Microcollapse, CIN, and Protein Concentration on Lyo Cake

Revealing the Pore Structure Variance of Lyophilized Cakes from Microcollapse, Controlled Ice Nucleation, and Protein Concentration Changes
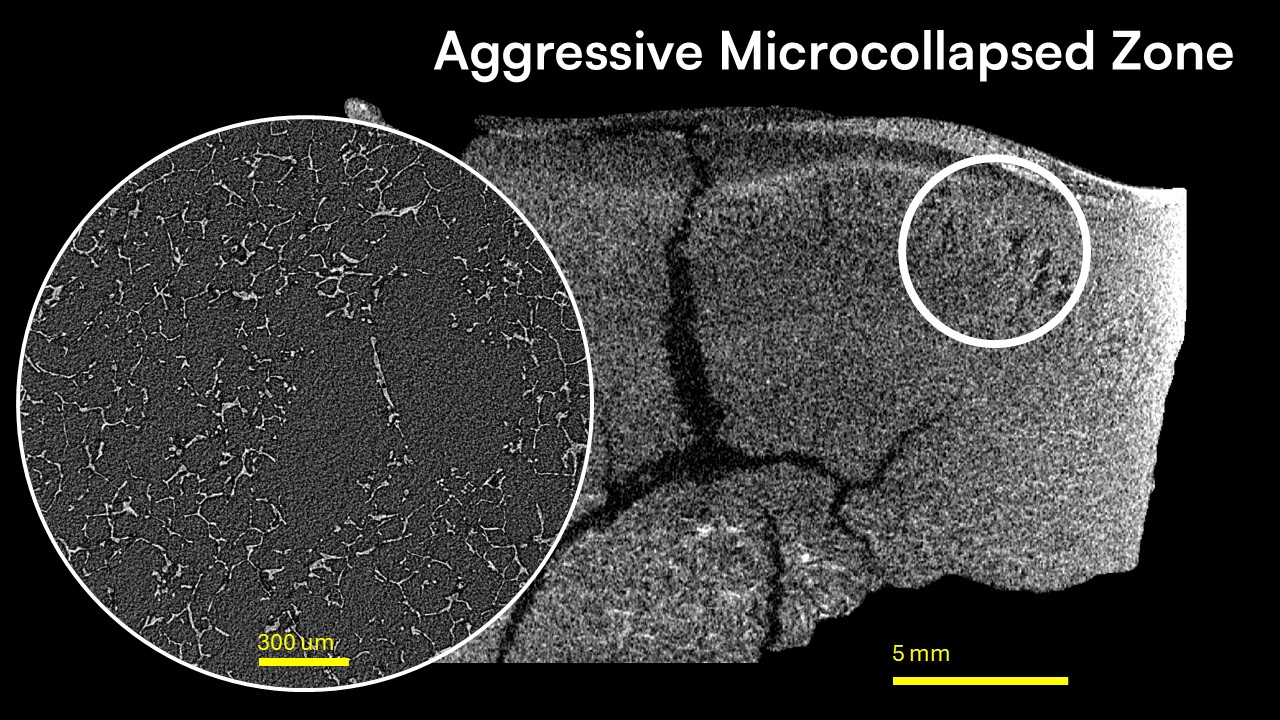
This study investigates the application of X-ray microscopy (XRM) as an advanced characterization tool for optimizing lyophilization cycles in pharmaceutical formulations. Utilizing non-invasive, high-resolution imaging, XRM provided comprehensive insights into the macro- and microstructural features of lyophilized cakes, revealing distinct differences attributable to lyophilization processing. High primary drying temperatures and aggressive conditions led to microstructural alterations, including fractures and microcollapse, while conservative drying methods resulted in more uniform and homogeneous structure. The controlled ice nucleation (CIN) process notably enhanced sublimation rates by forming extensive macroporous zones.
Calibrated intensity analysis via XRM detected variations in solid content, and high-resolution imaging highlighted subtle microstructural differences among samples, contributing to a deeper understanding of cake properties. Surface area measurements obtained from XRM showed a strong correlation with conventional BET analysis, validating its quantitative accuracy. Additionally, simulated diffusivity derived from XRM data correlated well with dry layer resistance values from thermocouple measurements, underscoring its utility in evaluating mass transport behaviors. XRM's non-destructive nature and detailed imaging capabilities make it a valuable complementary tool to existing characterization techniques, enabling more efficient cycle development and optimization.
Lisa Ma, Lokesh Kumar, Fred Lim, Shawn Zhang, Jeffrey Wong, Phil Yawman, and Debby Chang
In collaboration with the Genentech Inc. Pharmaceutical Development Department
Additional Publications
Transform Your Program with Microstructure Science
Get started with a drug product digital twin.














